
Caudal Epidural In Paediatric Population: Critical Analysis
Introduction
Caudal epidural blockade in children is one of the most widely administered techniques of regional anaesthesia. Caudal blocks are an efficient way to offer perioperative analgesia for painful sub-umbilical interventions with benefits of early ambulation, periprocedural haemodynamic stability and maintenance of spontaneous breathing in patients at maximum risk of a difficult airway .These are important advantages over general anaesthesia, notably in preterm babies and in children with cardiopulmonary co-morbidities.
Recent clinical studies have answered major pharmacodynamic and pharmacokinetic questions, thus providing the scientific background for safe and effective blocks in daily clinical practice and demonstrating that patient selection can be expanded to range from extreme preterm births up to 50 kg of body weight. The same developments have made it possible to reduce the dose concentrations of drugs used for caudal blocks..
Keywords: caudal block, paediatric, perioperative care, postoperative pain, ultrasound-guided.
Historical consideration
The first author to describe caudal anaesthesia in paediatric patients was Meredith Campbell in 1933. Over time, this idea has developed into a technique of great interest, especially for use in premature infants and in newborns, considering that these paediatric subgroups at high risk of perioperative respiratory depression are, as a result of an immature state of the CNS. In 1992 ,the first major publication from a single centre, including a cohort of 1100 children confirmed the reliability of the method which was reported by Veyckemans and colleagues . They were also the first to correlate complication rates to anaesthetist’s experience, concluding that the technique was easy to perform even at the beginner level.(1,2,3)
Nearly one-quarter of anaesthetic procedures which are today performed on children involve regional anaesthesia. Among them a single-injection caudal blocks are accounting for 34–40% of patients in paediatric regional anaesthesia. Based on central blocks, their share ranges from 80% in European centres up to 97% in the USA. According to data from the two largest multicentre studies available on the incidence and morbidity of regional anaesthesia in paediatric patients, caudal blocks are most commonly administered to children in the age range of 12 months up to 3 years.(4, 5)
Indications, contraindications and pitfalls
Sub-umbilical vs mid-abdominal indications
Caudal anaesthesia is indicated for surgical and non-surgical painful interventions in body areas below the sub-umbilicus which include procedures such as inguinal hernia repair, cystoscopy/transurethral manipulation, circumcision, anal atresia, treatment of limb ischemia, treatment of intussusception, or cast application to immobilise newborns with hip dysplasia. However, clinical experience has shown that caudal anaesthesia when used for mid-abdominal surgical interventions such as umbilical hernia repair has limited and unpredictable success rate. Reasons for this shortcoming might be age-dependent differences in the levels of sensory analgesia achievable by caudal blockade, and unpredictable secondary spread of local anesthetics.(6, 7)
Contraindications vs low-risk patients with spinal anomalies
Contraindications to caudal anaesthesia in children would include local site infection, pilonidal cyst, or spinal dysraphism such as tethered cord syndrome. In the presence of other spinal/meningeal anomalies, conducting a preoperative anatomical investigation by ultrasound or MRI is suggested. A careful risk-benefit analysis can help to identify patients at low risk of inadvertent nerve lesions, who might benefit from regional instead of general anaesthesia despite their anomaly (e.g. children with a difficult airway or preterm infants with a history of respiratory depression episodes). Any caudal blocks in these specific patients should be performed with ultrasound guidance and only by anaesthetists highly experienced with this technique.(1)
Simultaneously,a thorough preanesthesia evaluation is required to rule out any congenital coagulation disorders or therapeutic anticoagulation. Preoperative laboratory testing is indicated only if the patient or any of his or her family members have a positive bleeding history. (1)
Anatomical considerations
Access for caudal anaesthesia
A thorough knowledge and understanding of anatomical characteristics is key to the success of caudal blockade. Infants differ from adults with regard to their sacral anatomy and fat distribution.1,2 Due to of the individual variability of the developmental fusion processes through which the sacral vertebrae and ligaments undergo during childhood, the anatomy of the sacrum is highly variable. The epidural space can be entered through the sacral hiatus. Palpating structures for identification may be difficult if the cornua are of substantial thickness, and accessing the epidural space may be difficult in older children if the sacrococcygeal membrane cannot be penetrated because of an advanced stage of ossification. In terms of pharmacologically caudal blocks are both feasible and can be safely applied in children up to 50 kg of body weight.(1,3)
Age and body positioning
At what segmental level the spinal cord and dural sac terminate in a given patient will vary with both age and body positioning. Regarding age, the pubertal growth spurt has been found to involve cranial movement of the spinal cord termination from the L3 to a L1−L2 level within only 12 months. In contrast, Shin and colleagues observed in a study population of children that the dural sac ended lower than S2—S3 in 8% of patients. (7 )This lower position of the spinal cord and dura increases the risk of inadvertent dural puncture in newborns and toddlers. As to body positioning, Koo and colleagues demonstrated that lateral placement of patients with their neck, hips, and knees maximally flexed was associated with significant cephalad shifting of the dural sac. In other words, finding the right position for a patient can help to avoid complications.(8)
Palpation for needle insertion
Placing the patient in the left lateral decubitus position and the hips and knees flexed, the sacral hiatus can be identified using either the conventional landmark technique or ultrasound (Fig. 1). First of all, the posterior superior iliac spines are palpated via anatomical landmarks, the line drawn joining these two points represents the base of an equilateral triangle the tip of which indicates the position of the sacral hiatus. The sacrococcygeal ligament can be palpated between the two sacral cornua, through which the needle should penetrate the skin at an approximate 45° angle. Once the ligament has been passed, a flatter angle is adjusted by descending the needle before it can be advanced to the correct final position. It should be noted that Tuffier’s line does not seem to be an adequate reference point in neonates placed in a lateral flexed position, as it shifts dura to a significantly more caudal position . Before the local anaesthetic can be applied, cautious aspiration or passive drainage is required to rule out an inadvertent intravascular or spinal needle location.(1,3,4)
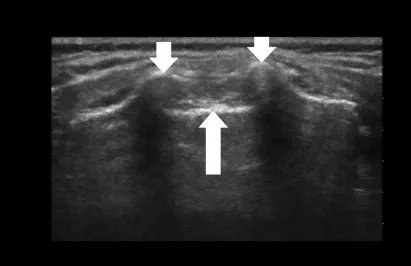
Technical considerations
Ultrasound vs landmarks
The landmark-based approach to caudal anaesthesia yields convincing success rates but also prone to block failure with the result of inadequate anaesthesia.(5) Ultrasound guidance, by comparison, offers two key advantages: it helps to identify small anatomical structures, and allows the spread of the local anaesthetic to be seen Fig 2 and 3.(1, 4,7) Ultrasound guidance does not need any special requirements on patient positioning or an additional aseptic technique. A sterile preparation of the ultrasound probe is obligatory. Fig 2, 3, 4 illustrate how a high-frequency linear-array transducer with a sterile cover is placed longitudinally in a position slightly paramedian to the lumbar spine and how the spread of local anaesthetic can be seen with this technique.(1) The superiority of ultrasound-guided puncture is obvious and unchallenged, especially in preterm babies and in infants whose sacral anatomy is not well understood. Yet no data from large-scale prospective studies are currently available to confirm that ultrasound offers better results and long-term outcomes in children of any age group managed by caudal anaesthesia. (7 )

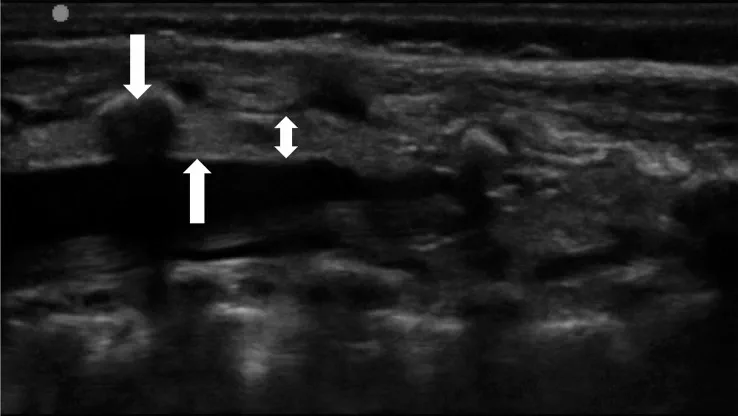
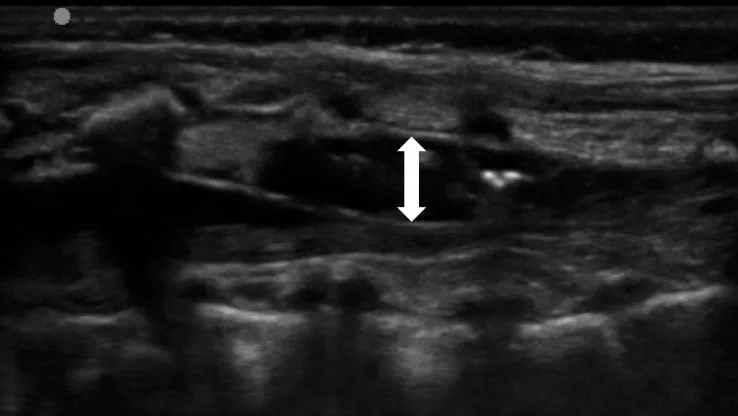
The ‘whoosh’ and ‘swoosh’ tests
Most single-injection caudal blocks in children take place without any technical aids or imaging.(5) Ultrasound guidance, although devoid of side-effects, was used in only 3% of patients. Landmark-based palpation of the sacral hiatus cannot guarantee that a needle is correctly inserted to its target position. Known risks of are accidental puncture of the dura or intravascular access. The ‘whoosh’ test was first described by Lewis and colleagues in a 1992 report involving adult patients as a specific and sensitive method to confirm needle placement in caudal anaesthesia.22 Injecting 2 ml of air into the epidural space resulted in a ‘whoosh’ sound, which was verified through a stethoscope kept over the thoracolumbar spine. After reports of neurological and haemodynamic side-effects from this air injection, the method was refined by injecting not air, but local anaesthetics and accordingly labelled as the ‘swoosh’ test.(8,9 )
Ultrasound vs swoosh/whoosh testing
Both the ‘whoosh’ and the ‘swoosh’ test proved to be similarly reliable when properly used. Still a major limitation of both tests is the subjectivity of examiners in judging the correct noise. Raghunathan and colleagues presented data affirming hypothesis that ultrasound was the superior method. They found that the best single indicator of successful epidural injection was to visualise by ultrasound in real time the turbulence generated by the local anaesthetic within the caudal space.. Guay J.et al concluded in a recent Cochrane Review that ultrasound improves the success rate of blocks and increases duration of block , especially in young children.(8,9 ,10)
Training and equipment requirements
Learning curve
Caudal blocks in children are easy to learn. Schuepfer G et al while studying learning curve amongst novice anaesthesia residents for paediatric caudal blocks observed that residents who had no prior experience in paediatric anaesthesia or in performing caudal blocks were found to require only 32 blocks for an 80% success rate. The learning curve started out in a steep increase, followed by a slight flattening once 15 blocks had been performed. Success rates of these untrained residents were comparable with those of experienced paediatric anaesthetists. (11)
Needle types
Cannulae for single-injection caudal blocks are available in different types and sizes, including a range of narrow-gauge (22- up to 25-gauge) short-bevel Tuohy and Crawford needles .Tissue trauma decreases with the calibre of the needle. Till now, paediatric caudal blocks are most commonly performed with needles that do not feature a stylet. (1)
Spreading of epidermoid tissue?
Hollow needles might increase the risk of epidermoid tumours after lumbar puncture because of tissue coring into the spinal space. However, the incidence of acquired epidermoid tumours is so low and only sporadic cases are reported. The lower risk of accidental spinal puncture in caudal limits the development of epidermoid. (12)
Complications and safety
Caudal blocks are known to involve haemodynamic/systemic or local adverse events. Examples include arrhythmia, hypotension when combined with general anaesthesia, respiratory depression resulting from inadvertent expansion of anaesthetics, toxicity-related seizures, infection/inflammation of the puncture site, sacral osteomyelitis, or local nerve injury. Yet, the morbidity associated with any of these events is low. Ecoffey and colleagues analysed data of 31,132 regional anaesthetic procedures and identified only eight patients with complications related to caudal blocks: six dural taps (without postdural puncture headache), one nerve injury, and one case of cardiac toxicity. Polaner and colleagues even reported ‘no complications in the caudal group’, pointing out that the main periprocedural problems were inability to place the block and block failure. (13,14,)
Choice of local anaesthetics
Bupivacaine vs ropivacaine
Common drugs for caudal blockade are (levo)bupivacaine 0.125–0.25% and ropivacaine 0.1–0.375%, used at a volume of 0.5–1.5 ml kg−1 depending on the desired dermatomal level. Current guidelines recommend that doses should not exceed 2 mg ml−1 for ropivacaine and 2.5 mg ml−1 for bupivacaine, and the recommended volumes are 0.5 ml kg−1 when sacral dermatomes, 1.0 ml kg−1 when lumbar dermatomes, or 1.25 ml kg−1 when lower thoracic dermatomes are achieved. Ropivacaine is known to cause less postoperative motor blockade than bupivacaine. (14,15)
Toxic events and their management
Systemic toxic events from local anaesthetics may involve cardio- or neurotoxicity. Current guidelines recommend that any haemodynamic deterioration be treated by Intralipid® 20% as first-line therapy along with epinephrine/adrenaline for cardiopulmonary resuscitation until circulation is restored or extracorporeal membrane oxygenation has been installed. The Intralipid® should be administered in these situations i.v. as a rapid bolus injection of 1–1.5 ml kg−1 followed by continuous infusion (0.25 mg−1 kg−1 min−1) and repeated bolus every 3–5 min up to 2–5 (−10) ml kg−1. Some regimens make the administration of a maintenance fluid (0.25 ml kg−1 min−1) seem useful. Neurotoxic seizures need to be treated with propofol, benzodiazepines, or barbiturates. (1,14)
Use of adjuvant drugs
Preservative-free morphine and clonidine are registered drugs for epidural use. Any of the other drugs listed below are administered widely, but off label in this context. The very latest recommendations issued by the European/American Society of Regional Anaesthesia endorse the use of alpha-2 agonists (clonidine, dexmedetomidine), preservative-free morphine, and ketamine as adjuvants in caudal blocks. (1,14)
Volume dosing and cranial spread
Dermatomal reach
Both to ensure an adequate level of analgesia during caudal blockade and to avoid side-effects, it is essential to calculate the proper amount of local anaesthetic. A number of confounders affecting the cranial spread of local anaesthetics include body weight, body height, age, and injection speed. Weight-based formulas have a long tradition in paediatric regional anaesthesia but one also has to consider the desired reach of a block in terms of dermatomal level. Epidural space volume is known to increase continuously from caudal to cranial. A recent study has revealed median volumes of 1.30, 1.57, and 1.78 ml kg−1 at the L1, T10, and T6 levels, respectively. Hence current guidelines continue to recommend the well-established formula—introduced by Armitage in 197 where 0.5 ml kg−1 is expected to reach sacral, 1.0 ml kg−1 lumbar, and 1.25 ml kg−1 mid-thoracic dermatomes.(15,16)
Cranial rebound
Several studies have relied on ultrasound to investigate the cranial spread of local anaesthetics in paediatric caudal blocks.6, 7, 62 The reach of high-volume blocks was found to be inversely related to age.64 Radiography and ultrasound revealed that, no matter how much of a local anaesthetic was used, its cranial spread never seemed to reach past the T-10 level immediately upon injection.62, 63 In contrast, skin testing revealed T-4 dermatomal levels within several minutes of carrying out the injection. Lundblad and colleagues7 discovered a rebound mechanism of CSF behind this phenomenon. In phase I of this mechanism, CSF recedes in the direction of the cranium as epidural pressure is rapidly building up during injection of the local anaesthetic. (15,16,17)
In phase II, physiological interactions between intracranial and spinal pressure gradients cause the CSF to return in a caudal direction, thus forcing in its stead the epidural bulk of local anaesthetic to flow in the direction of the cranium. This secondary spread of the drug covers a distance of approximately two more spinal-cord segments within 15 min of injection.(7)In contrast to relative pressure changes, the speed of injection does not affect the cranial spread of local anaesthetics. Weight-based formulas may promise of ensuring intraoperative analgesia, but, in daily clinical practice, caudal blocks are occasionally inadequate even at dose concentrations high enough.(15,16,17)
Caudal approach for lumbar and thoracic anaesthesia
While the term ‘caudal block’ is mainly used of single-injection procedures, a caudal approach can also be taken for lumbar and thoracic epidural catheterisation. In clinical practice, however, this practice is so uncommon that only 1% of children were managed by caudally inserted catheters in the aforementioned large-scale multicentre study of European provenance.(4) Two major risks are of concern. The first one concern includes the high risk of caudal catheters getting bacterially contaminated in this environment of nearby excretory organs. Subcutaneous tunnelling of the catheter or a slightly higher insertion point (L-5/S-1 in a ‘midline modified Taylor approach’) can help to control the risk of infection. The second issue concerns the requirement to verify correct placement of the catheter, with regard to both an adequate spinal-cord level and the epidural position. Failure rates of 20–30% in epidural catheter tip placement have been reported Epidurogram remains gold standard for correct catheter tip placement but not feasible .(20,21,22)
Conclusion
Despite the growing popularity of abdominal wall blocks in recent years amongst paediatric patients, a single shot caudal epidural is still preferred by most anaesthesiologists for infraumbilical surgeries and non surgical interventions.
References:
1. Marion Wiegele,Peter Marhofer, and Per-Arne Lönnqvist . Caudal epidural blocks in paediatric patients: a review and practical considerations. Br J Anaesth. 2019 Apr; 122(4): 509–517.
2. Polaner D.M., Taenzer A.H., Walker B.J. Paediatric Regional Anesthesia Network (PRAN): a multi-institutional study of the use and incidence of complications of pediatric regional anesthesia. Anesth Analg. 2012;115:1353–1364. [PubMed] [Google Scholar]
3. Lundblad M., Lönnqvist P.A., Eksborg S., Marhofer P. Segmental distribution of high-volume caudal anesthesia in neonates, infants, and toddlers as assessed by ultrasonography. Paediatr Anesth. 2011;21:121–127. [PubMed] [Google Scholar]
4. Ecoffey C., Lacroix F., Giaufré E., Orliaguet G., Courrèges P. Association des Anesthésistes Réanimateurs Pédiatriques d’Expression Française (ADARPEF). Epidemiology and morbidity of regional anesthesia in children: a follow-up one-year prospective survey of the French-Language Society of Paediatric Anaesthesiologists (ADARPEF) Paediatr Anaesth. 2010;20:1061–1069. [PubMed] [Google Scholar]
5. Jöhr M. Regional anaesthesia in neonates, infants and children: an educational review. Eur J Anaesthesiol. 2015;32:289–297. [PubMed] [Google Scholar]
6. Keplinger M., Marhofer P., Klug W. Feasibility and pharmacokinetics of caudal blockade in children and adolescents with 30-50 kg of body weight. Paediatr Anaesth. 2016;26:1053–1059. [PubMed] [Google Scholar]
7. Shin S.K., Hong J.Y., Kim W.O., Koo B.N., Kim J.E., Kil H.K. Ultrasound evaluation of the sacral area and comparison of sacral interspinous and hiatal approach for caudal block in children. Anesthesiology. 2009;111:1135–1140. [PubMed] [Google Scholar]
8. Koo B.N., Hong J.Y., Kim J.E., Kil H.K. The effect of flexion on the level of termination of the dural sac in paediatric patients. Anesthesia. 2009;64:1072–1076. [PubMed] [Google Scholar] 11. Orme R.M., Berg S.J. The ‘swoosh’ test—an evaluation of a modified ‘whoosh’ test in children. Br J Anaesth. 2003;90:62–65. [PubMed] [Google Scholar]
9. Schwartz D., Raghunathan K., Han D. The ‘Doppler-swoosh’ test: a further modification to the ‘swoosh’ test. Paediatr Anaesth. 2007;17:600–601. [PubMed] [Google Scholar]
10. Raghunathan K., Schwartz D., Connelly N.R. Determining the accuracy of caudal needle placement in children: a comparison of the swoosh test and ultrasonography. Paediatr Anaesth. 2008;18:606–612. [PubMed] [Google Scholar] 1
11. Schuepfer G., Konrad C., Schmeck J., Poortmans G., Staffelbach B., Jöhr M. Generating a learning curve for pediatric caudal epidural blocks: an empirical evaluation of technical skills in novice and experienced anesthetists. Reg Anesth Pain Med. 2000;25:385–388. [PubMed] [Google Scholar]
12. Morita M., Miyauchi A., Okuda S., Oda T., Aono H., Iwasaki M. Intraspinal epidermoid tumor of the cauda equina region: seven cases and a review of the literature. J Spinal Disord Tech. 2012;25:292–298. [PubMed] [Google Scholar]
13. Flandin-Blety C., Barrier G. Accidents following extradural analgesia in children. The results of a retrospective study. Paediatr Anaesth. 1995;5:41–46. [PubMed] [Google Scholar
14. Suresh S., Ecoffey C., Bosenberg A. The European society of regional anaesthesia and pain therapy/American society of regional anesthesia and pain medicine recommendations on local anesthetics and adjuvants dosage in pediatric regional anesthesia. Reg Anesth Pain Med. 2018;43:211–216. [PubMed] [Google Scholar]
15. Suresh S., Kopp S. The use of ultrasound guidance for perioperative neuraxial and peripheral nerve blocks in children: a Cochrane Review. Anesth Analg. 2017;124:948–958. [PubMed] [Google Scholar]
16. Armitage E.N. Caudal block in children. Anaesthesia. 1979;34:396. [Google Scholar]
17. Brenner L., Marhofer P., Kettner S.C. Ultrasound assessment of cranial spread during caudal blockade in children: the effect of different volumes of local anaesthetics. Br J Anaesth. 2011;107:229–235. [PubMed] [Google Scholar]
18. Triffterer L., Machata A.M., Latzke D. Ultrasound assessment of cranial spread during caudal blockade in children: effect of the speed of injection of local anaesthetics. Br J Anaesth. 2012;108:670–674. [PubMed] [Google Scholar]
19. Thomas M.L., Roebuck D., Yule C., Howard R.F. The effect of volume of local anesthetic on the anatomic spread of caudal block in children aged 1–7 years. Paediatr Anaesth. 2010;20:1017–1021. [PubMed] [Google Scholar]
20. Bosenberg A.T., Bland B.A., Schulte-Steinberg O., Downing J.W. Thoracic epidural anesthesia via caudal route in infants. Anesthesiology. 1988;69:265–269. [PubMed] [Google Scholar]
21. Kost-Byerly S., Tobin J.R., Greenberg R.S., Billett C., Zahurak M., Yaster M. Bacterial colonization and infection rate of continuous epidural catheters in children. Anesth Analg. 1998;86:712–716. [PubMed] [Google Scholar]
22. Bubeck J., Boos K., Krause H., Thies K.C. Subcutaneous tunneling of caudal catheters reduces the rate of bacterial colonization to that of lumbar epidural catheters. Anesth Analg. 2004;99:689–693. [PubMed] [Google Scholar]
By Dr. Vrishali Ankalwar
Associate Professor
GMCH, Nagpur
